Hot Sales
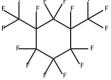
Perfluoro-1,3-dim...
|
DOPA-Phe-Phe-OH
|
Ethyl...
|
(2R,3R,4S,5R)-2-(...
|
1-(Benzo[d][1,3]d...
|
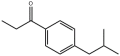
1-(4-Isobutylphen...
|
(2-ethylbutyl...
|
(S)-2-ethylbutyl...
|
Recent Browse
Parameters
| Properties | |
| Properties | 【熔点 】60-62 °C(lit.)【闪点 】>230 °F【储存条件 】−20°C |
|---|---|
| Specification And Standard | |
| Production And Usage | |
| Production process | (R,S)-2-Hydroxyhexanoic acid (1h): General procedure for the preparation of alpha-hydroxycarboxylic acids (GP1): In a round bottom flask, 100 mmol alpha-amino acid was dissolved in 196 mL (200 mmol) of 1 N sulfuric acid. The solution was cooled to 0 degC and added with a cooled solution of 13.8 g (200 mmol) sodium nitrite in 30 mL water. The reaction mixture was stirred for 3 hours at 0 degC and then at rt overnight. Thereafter, the solution was heated for 20 min to 60 degC and after cooling extracted with diethyl ether (3 x 100 mL). The organic phase was separated, dried over Na2SO4 and the solvent was removed under vacuum. The product was further used without additional cleaning. From 13.2 g (100 mmol) D,L-norleucine in 196 mL (200 mmol) of 1 N H2SO4 and 13.8 g (200 mmol) of NaNO2, according to GP1, (R,S)-2-hydroxyhexanoic acid was obtained as a yellow oil. The compound was used without further purification. Yield: 10.44 g (79 mmol, 79 %). |
| Packing And Storage | |
| Safety | |
| Spectrum And References | |
| Contact Salesman | |
| Buy reagents | 400-808-6996 |
| Update | 20200220marketing |



![(2R,3R,4S,5R)-2-(4-aminopyrrolo[2,1-f][1,2,4]triazin-7-yl)-3,4-dihydroxy-5-(hydroxymethyl)tetrahydrofuran-2-carbonitrile](http://www.chemvia.com/images/goods/20221206/83f151cec8f44578.jpg)
![1-(Benzo[d][1,3]dioxol-5-yl)propan-1-one](http://www.chemvia.com/images/goods/20221206/97d2d867bb9df4d8.jpg)